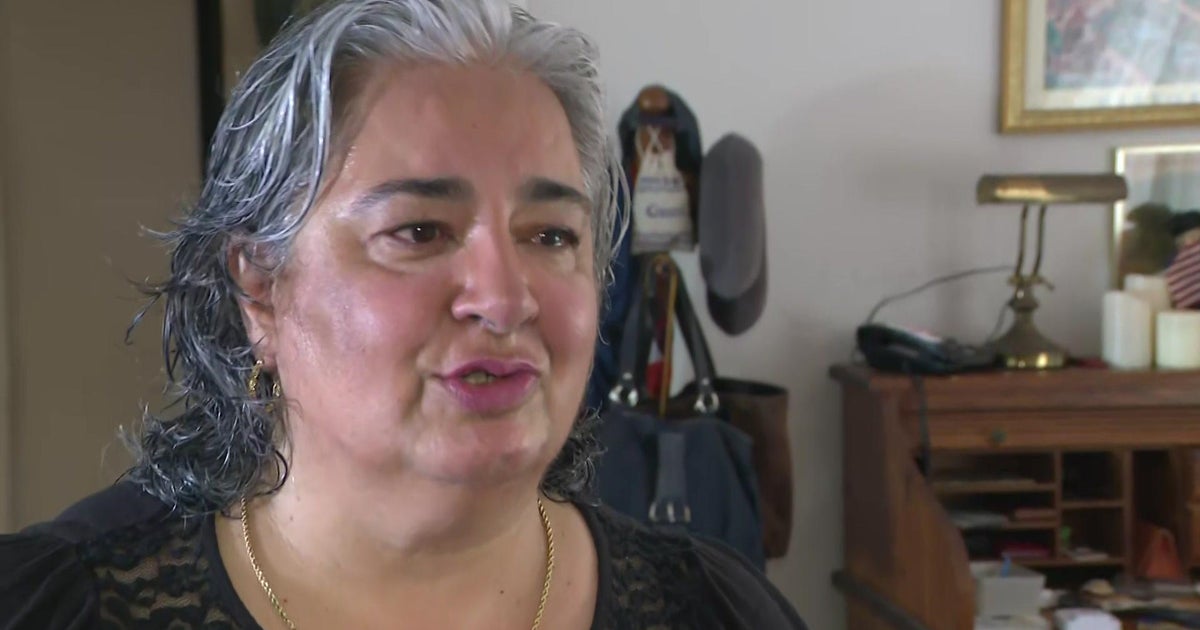Exclusive: FDA Sues Sunrise Stem Cell Clinic After Several Patients Blinded, Sustain Eye Damage
Follow CBSMIAMI.COM: Facebook | Twitter
SUNRISE (CBSMiami) – The Food and Drug Administration is taking a South Florida stem cell clinic to court, alleging that the clinic is "experimenting on patients with adulterated and misbranded drugs" and refuses to obey the law.
The clinic, U.S. Stem Cell, has also faced scrutiny after several patients say they were blinded or suffered significant vision loss following treatments at the facility.
Jeannine Mallard is one of those patients. She recently spoke to CBS4 News and said she flew from her home in France to South Florida in 2015 to get treatments for macular degeneration. Clinical trials can be inherently risky.
"I could not wait doing nothing and I expected this to improve," she said.
Mallard says she came to the Hollywood Eye Institute clinic in Cooper City to be treated by Kristin Comella and Dr. Shareen Greenbaum. Mallard paid several thousand dollars to participate in a clinical trial run by a company called Bioheart, the precursor to US Stem Cell.
"They said it was sure," Mallard said. "They said the worst that could happen is there would be no effect so I could not imagine what would happen next."
What happened next, according to Mallard and her attorney Andrew Yaffa, is a cautionary tale. Immediately after the procedure Mallard began to experience inflammation and pain.
According to the Mallard and Yaffa, the procedure involved taking Mallard's stem cells from her fat tissue, treating the stem cells and injecting them directly into her right eyeball and behind her left eyeball. Mallard said Greenbaum reassured her about the pain she was having.
"She said it's just inflammation, to not last," Mallard said. "It's normal."
It wasn't normal. Mallard is now blind in her right eye and she suffered significant vision loss in her left eye. Her right eye is noticeably shrinking.
"It's becoming smaller," Mallard said. "So small it's dead right now."
CBS 4 News tried to reach Dr. Greenbaum through calls to her office, emails and a visit to her office. She did not respond to our requests.
And Mallard is not alone. Attorney Yaffa sued on behalf of two other women who underwent similar procedures at US Stem Cell.
Court records and a report in the New England Journal of Medicine show that those other patients and a fourth woman experienced similar significant vision problems. Court documents show that in those cases the parties settled the claims.
Yaffa alleges in his lawsuit and in an interview with CBS4 News that US Stem Cell preyed on Mallard who feared losing her vision to macular degeneration.
"This is human experimentation and they are exploiting patients that are so desperate and vulnerable that they are looking for any hope at all," Yaffa said.
The Food and Drug Administration is taking action. Last month, the FDA filed a lawsuit to have US Stem Cell stop marketing stem cell therapies that do not have FDA approval. The lawsuit says, "An injunction is necessary to prevent Defendants from experimenting on patients with adulterated and misbranded drugs."
The agency sent Kristin Comella at US Stem Cell a warning letter last year, saying the procedures the clinic is doing need to be regulated by the FDA like a drug.
The FDA says in this warning that unproven stem cell therapies need to be regulated because if cells are manipulated by a significant amount after their removed, there is a risk of contamination of the cells.
But in a series of online videos and speeches, Kristin Comella, the Chief Science Officer at U.S. Stem Cell, makes it clear she disagrees with the FDA.
"We have helped thousands of patients harness their own healing potential and it would be a mistake to limit these therapies for patients who desperately need them," Comella said.
Comella, who is not licensed medical professional according to the state of Florida and whose background, according to her online biography is in chemical engineering and regenerative medicine with advanced degrees in chemical engineering and stem cell biology, pitched stem cell therapies as a wonder cure, treating everything from serious brain and spinal cord injuries to hair loss. She touts numerous success stories.
"We've had patients who were wheelchair bound come out of their chairs," she said in an online video. "We've had patients who were unable to speak able now speaking full sentences telling us about the day they were injured."
But in her speeches there was little discussion of patients like Jeannine Mallard who suffered serious eye damage. We requested an on camera interview with Comella through her public relations firm but we were denied. So, we tracked Comella down in Weston to ask her about the allegations. She said she believes the government should not interfere in a patient's right to utilize their stem cells.
"It is our goal to protect the rights of patients to use their own stem cells that exist inside their body to promote healing," Comella said.
We asked her specifically about Jeannine Mallard, and others, who suffered vision loss.
"These poor women cannot see," a reporter said. "They have significant vision loss and they trusted the clinic to do the work for them. Nothing to say to that?"
Comella did not respond and did not want to comment on the Mallard case, citing an ongoing lawsuit filed by Mallard's attorney. In fact, Comella didn't want to talk about much — including the FDA lawsuit and the work at U.S. Stem Cell.
"So, you don't want to answer these questions about the clinic and the work that's being done there and whether it's safe?" a reporter asked.
Comella responded, "I'm gonna ask you to meet with our PR people and continue to reach out to them for additional comments. I'm gonna ask you to turn the cameras off."
In a statement sent to CBS4 News from the PR company, Comella said, "My entire career has been dedicated to studying and developing treatments utilizing the body's own natural ability to heal, and I am devoted to helping others achieve optimal health through therapeutic procedures. It is my life's work to pioneer regenerative medicine and educate the public about its healing potential. I remain steadfast that no government agency should deprive individuals of their right to harness the cells that exist in their body."
The company also sent us a link to a page on the U.S. Stem Cell website directly responding to the FDA lawsuit. You can find that by clicking here.
Dr. Thomas Albini, Professor of Clinical Ophthalmology at Bascom Palmer Eye Institute, University of Miami, treated Mallard and co-authored the New England Journal of Medicine paper on 3 other patients from US Stem Cell that sustained permanent eye damage from the stem cell procedure. He believes much more study needs to be done before performing these treatments on people.
"I think it's really wrong to offer these types of therapies to people when they're very dangerous and people aren't aware of the risks involved," Albini said. "It's not that there's something wrong with stem cells or stem cells and eye disease. Stem cells may very well wind up being the cure for a lot of eye diseases. It was the way in which it was studied and the amount of clinical study that went into doing it before it was offered to patients."
Albini also offered several important pieces of advice for people who might be looking at stem cell therapies for treatment. One, if you enroll in a clinical trial you almost never have to pay for it. Patient funded research is seen as a conflict of interest in the medical field, and two, you should ask if the doctor treating you and recommending stem cell therapy has the appropriate medical background to treat the disease or condition you have.
"I think its paramount to require clinicians who are going to be doing these types of therapies to have conducted research ahead of time," Albini said.
According to the FDA, they have the authority to regulate stem cell products in the U.S. and in most cases, the agency works to "determine the safety and effectiveness of products in well-controlled human studies." Currently, the FDA says that the only stem cell products that they've approved for use in the U.S. are derived from cord blood.
The FDA suggests you ask your health care provider if the FDA has approved the stem cell treatment they are recommending for you before agreeing to it.
Jeannine Mallard says she learned of the US Stem Cell clinic through a clinical trial posted on the National Institutes of Health website. Mallard believed that gave the trial legitimacy.
There is a disclaimer on the site that the trials listed have not been vetted by the government but Mallard still believes it's wrong for it to be posted on a government website.
"What makes me really angry is to find this on the National Institutes of Health," she said. "How can this be listed on such official site, website? I still don't understand that."
Mallard says her life is vastly different now. She can't read and relies on friends and family for nearly everything. She recalls often the past two years of doctors in France trying to repair her eye and being unable to.
"They did their best but now they could not save it," she said.